
- Effizienz durch Wiederverwendung
- Konsistenz durch Vermeidung von Redundanz
- Organisatorische Herausforderungen


{fastsocialshare}
Last week I attended the Inartis Network seminar "E-connected Healthcare - Innovation Made in Switzerland in Digital Health and Telemedicine" that covered the latest developments in the eHealth market. The eleven speakers provided some very interesting input on their take on the problems and solutions in this domain. The common denominators were:
The seminar was utterly silent on the regulatory aspect of all these developments. There was only one point where this came up: as Mark-Eric Jones, the CEO of Leman Micro Devices was presenting the company's smartphone blood pressure solution, he got the question if this "did not make the smartphone a medical device"?
We can all imagine where this could lead. If the smartphone becomes a medical device then it needs to be developed and documented accordingly. Who would do this work? I cannot imagine that Apple and Samsung are particularly keen on the idea.
Mr. Jone's ingenious answer was to declare the smartphone as a medical device accessory "..like a battery. If your device needs a battery, you don't need a special medical device documented battery. You can take any battery as long as they fulfill the specifications you set up as a medical device manufacturer. It's the same with the smartphone. We define the required specifications and you can then use any smartphone that suits these specs."
Mr. Jones claimed that Lemans had worked closely with the regulatory authorities on this issue and had received acceptance of this approach provided that "additional software safety measures are put in place."
A very interesting approach. It remains to see if this is a viable approach for other manufacturers.
Request a live demo and let us show you how Aligned Elements can manage your documentation
{fastsocialshare}

"But it all becomes Class C?!?" - IEC 62304, software architecture and Software safety classification
During a seminar some time ago, I elaborated on the possibility of using existing risk assessments to deduce the software safety classification (SSC) of the software architecture parts according to IEC 62304 (see the slides here).
My discussion was based on the Matrix Model, initially published by the company "Certified Compliance Solutions". The essence of the deduction goes like this:
So how can the risks emanating from the functional context often documented in SW requirements, be transposed to the SCC of the Software Architecture.
The company “Certified Compliance Solutions” ties these concepts together by applying the so-called "Matrix model" to functional workflows and the software architecture. The approach is best explained in the picture below.
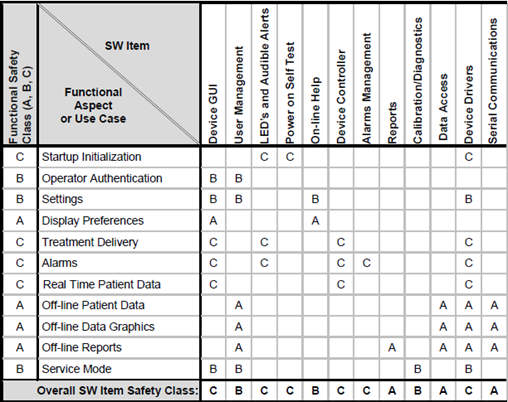
The idea is to establish/document use-cases for all SW Requirements and perform a risk assessment on these Use Cases to establish the worst possible harm they might cause.
This gives each Use-case a "functional" Safety Classification.
On the X-axis, we enumerate all Software Items/Units in our architecture.
It should now be possible to single out the set of Software Items/Units a particular Use-case needs/uses for its implementation.
The functional safety class for the Use-case can then be transferred to all the Software Items/Units needed for its implementation. Repeat this process for all Use-cases.
The highest occurring Software Safety Classification in each column represents the Software Safety Classification for the Software Item/Unit of that column.
(Be aware: A Use-case usually does not cause the same risk for each step of the workflow. Some steps generate higher risks than others. The Matrix Model does not account for this.)
"But then all becomes of Class C?!?" - exclaims a young man in the audience.
Now, this is a valid observation and a phenomenon we see from time to time.
Risk-driven development is one of the underlying reasons for IEC 62304. The manufacturer should focus development efforts on the software parts that pose the highest risks.
The Software Safety Classification-concept is used to detect these software parts. More risk => more effort required to ensure safety.
However, manufacturers sometimes seem overburdened with the ins-and-outs of the regulation and therefore capitulates to the approach of "just smack class C (or class B) on everything and get it over with".
I think there are several reasons for ending up in this situation and these reasons should not be confused with each other. They each have different causes and different remedies.
The first potential reason lies in the selected software architecture.
If, after having applied the Matrix Model, it turns out that all Software Items/unit indeed receive the highest possible software safety class, then this is might indicate a particularly entangled design with insufficient separation of concerns.
The Matrix Model should give an indication of high-risk Use Cases that span across wide parts of the architecture.
The scope of these use cases might be too large. Reconsidering the scope and aiming for a more risk-driven division of the workflows could lead to some important decomposition gains.
Or maybe the decomposition of the architecture is insufficient. Further decomposition and/or segregation of software items can potentially confine high-risk aspects of the software to dedicated areas.
A second and completely different reason for "everything turning into class C" lies in the administrative overhead of separating the work needed for each Software Item according to its Software Safety Classification. This point-of-view advocates that it is simply less work to treat every software item uniformly from an administrative perspective regardless of the actual risk of the software items is lower than nominally designated risk.
This indicates that something is foul in the development process and/or documentation system. IEC 62304 gives the manufacturer the possibility to perform efficient development, allowing redistribution of resources from low-risk areas to high-risk areas of the software. That opportunity is foregone in this scenario.
A well-adapted development process and supporting tool-set shall make sure that the following things are in place:
The third reason for this phenomenon is, of course, that the result is entirely warranted. The Matrix Model might actually correctly show that the entire software DOES carry a risk for death or serious injury to the patient and/or user. If this is the case, the model has served its purpose.
Last but not least, your development process might be more ambitious than what IEC 62304 requires. This is entirely OK. IEC 62304 establishes the most basic requirements to ensure that the software is developed with safety in mind. Raising the bar further by applying even more rigorous development requirements with regards to safety might very well be warranted for companies that put safety and quality first.
Learn more about how Aligned Elements can help you support IEC 62304
Request a live demo and let us show you how Aligned Elements can help you to comply to IEC 62304
{fastsocialshare}

To accelerate your development documentation process, Aligned Elements supplies a number of free downloadable extensions.
These include:
The extensions can be used in your existing projects to speed up the documentation work, to serve as inspiration, or to be used as enhanced control mechanisms.

For those considering using electronic instructions for use (eIFU) according to EU 207/2012, take a closer look at the "Electronic Instructions For Use Checklist (EU 207/2012)" that helps you finding out whether your device and its intended use makes is it qualified for the regulation.
The QA-based checklist provides objective evidence, once completed, that you have made a careful and detailed analysis of your device according to EU 207/2012.
The advantages of eIFUs are many and compelling, including:
Use this checklist to confirm that the regulation applies to your device.
If the checklist establishes that EU 207/2012 applies to your device, proceed and download the 38 predefined eIFU Requirements extracted from the EU 207/2012 and import them into your project.
Accelerating the development documentation work could not be easier!
{fastsocialshare}

As an example of Aligned Elements project consistency control, the Aligned Elements Pre-Submission checklist demonstrates how various aspects of the project content can be analyzed in order to avoid failed audits and unwelcome FDA 483 warning letters.
Both EN/ISO 13485 and EN/ISO 14971 are concerned with the completeness and consistency of the project content and this checklist serves as an instrument to make that effort easier and faster.
The checklist guides the user through a number of analysis action steps and optionally records the results in a checklist report, which serves as objective evidence of the analysis. Detailed instructions and screenshots accompany the steps to facilitate the execution.
The checklist has been designed to check a project that uses the Aligned Elements default templates.
The checklist verifies test points such as:
The checklist further performs optional checks, if you have selected to use features such as:
The Checklist, which is implemented as a Regulatory Wizard, is available for Aligned Elements users on the Extensions page in the Wizard section.
{fastsocialshare}

"If it is not documented, then it has not been done", according to the FDA saying. "Documentation not available", or "Documentation not adequate" are the most frequently cited deviations in FDA Warning Letters. The effects of inconsistent documentation can be devastating, implying postponed market launches, product withdrawals, and fines.
The reason for this flood of FDA 483 warning letters, addressing seemingly obvious and simple errors, is not that the medical device manufacturers are ignorant or incompetent.
It is simply hard to keep the DHF documentation consistent.
The documentation requirements are many and detailed, the development projects often span over a long time-period, involves a large number of alternating team members, all contributing to the large set of deliverables that make up the Design History File/Technical File. The deliverables are highly interdependent and a small change can cause unexpected ripple-effects over large parts of the documentation. A considerable amount of the total project effort is thus placed in the handling and management of the DHF.
Not long ago, I was contacted by a customer. He had recently taken over a project with the objective to take the product to market. After a brief introduction, he found the project to be in a miserable state. The project had switched project manager four times during the last four years, the team members were all new, knowledge about the documentation process was lacking.
In short, the situation was very opaque.
The project manager wanted help with assessing the current state of the development documentation and within 10 minutes, we could extract the following information from Aligned Elements:
The project manager finally felt that he had some grip on the situation. He now had concrete errors to fix and also the names of the people to contact to get detailed information about each individual inconsistency.
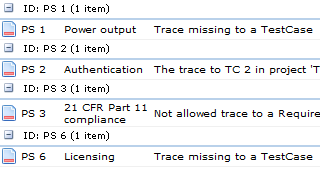
Aligned Elements addresses the issue of incompleteness with a range of integrated and automatic consistency and control functions. Aligned Elements is able to:
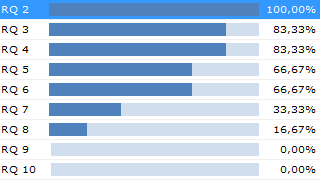
By the application of configurable validation rules, real-time checks can be executed on the documentation continuously. Faults and gaps are uncovered well in time before the auditor arrives or before the documentation is submitted to the notified body.
Knowing the state of the development documentation is invaluable for a medical device manufacturer. The list of open errors, representing the project’s “Documentation Quality Debt”, is an excellent estimator for how much work remains until the documentation is ready for release.
Reducing project risk by making the current documentation state transparent is an excellent way to increase the chances of a successful product launch.
Schedule a live demo and let us show you how Aligned Elements keeps your DHF complete and consistent
{fastsocialshare}
{fastsocialshare}
It is not hard to guess that Mobile Medical Applications (MMA:s) are going to be a hot topic for 2015. Just in time for Christmas, the market is flooded with wearables and connected devices for health-related applications. It is also predicted that we will see rapid growth in this area, bringing in a whole new group of companies (mobile app makers) into the medical device world.

From a documentation point of view, there might be some confusion on what really counts as an MMA. FDA has put together a great set of information, explaining their take on the subject with some reasonable helpful tips on how they will make their judgment. I recommend that you take a look.
Learn more about how Aligned Elements can help you to manage your medical device documentation.
Or contact us for a live demo.
{fastsocialshare}
Our friend Dr. Samuel Fricker, assistant professor at Blekinge Institute of Technology, is co-authoring the publication "Requirements Engineering for Digital Health".

According to the information, this book:
Samuel provided valuable help when we compiled our report on "Requirements Engineering for Medical Devices".
{fastsocialshare}

{fastsocialshare}
I was recently asked by a Business Developer at the Sahlgrenska Science Park Start-up incubator in Gothenburg, Sweden, about any useful medical device development documentation tips.
The concrete question was: "What is the absolute minimum a med-tech start-up must know about development documentation during the early phases in order to not suffer from mistakes later on?"
The development documentation essentially describes how you have developed the medical device.
This includes, but is not restricted to:
At product launch, all these things need to be in place, with all documents properly written, reviewed, signed off, and archived.
It is obvious to everyone involved that documentation means work. A lot of work. (We assess that up to 30% of the total development effort is spent on documentation). It is equally obvious that conducting this documentation work ties down resources that (due to mutual exclusion) is not spent on something else.
The practical effect of this is that once you start with the formal development (and documentation) process, everything else seems to slow down. And hence, there is often a question about "When does the formal development actually start?"
Now, while the regulations do not make any distinction between "formal" and "informal" development (there is only "formal" development), these start-ups are in such an early phase that they do not even know if there is going to be a product at all, or what the product should be.
What do companies in this situation have to know about development documentation?
Here is my advice:
Many of these rules are common sense and the sooner you start to use them, the better. Therefore, there is nothing preventing you from starting to use GDP right away and the extra work incurred is relatively low.
A good place to start is to get familiar with some of the most basic medical device development concepts and to have a good grip on these before the “formal development” starts. Even though these concepts have a limited influence on how you write documents during the early phases, knowing them well before formal development starts will make the transition from prototype to product a whole lot smoother.
I have listed some of the most important concepts below.
The QMS is a set of business processes that are meant to "assure product safety and efficacy". Essentially, it formally describes how your company performs these business processes (such as purchasing, manufacturing, development, service, HR, etc.) in order to make sure that the patient's safety is not at risk.
This is what you need to know:
Each geographical market has its own set of regulations. Even though the global community is working on harmonizing standards and norms, there are still local particularities.
Broadly speaking, regulations in the USA and China are considered more "difficult" to comply with whereas the EU and Canada are considered less "difficult".
Selecting target markets does therefore have a large impact on the development documentation required later on.
The "Intended Use" stipulates the use your product is intended for. This probably sounds straight-forward. However, I cannot emphasize enough the enormous impact the intended use has on the documentation burden lying ahead. Note that the intended use is not “what the device is designed for” or “what you can do with the device”. It is what you say your device is to be used for,
The classic example is the scalpel. If you stipulate the intended use to be “cutting tissue” in the general sense, then it is a Class I device according to the FDA. But if you label the exact same scalpel to be used for ophthalmology or eye surgery, then it is suddenly a Class III device.
Furthermore, according to the FDA, if your device is equivalent to a device already existing on the (US) market, then less work is needed to bring it to the market. The “equivalence” is largely decided based on the intended use.
Therefore, the way you word the intended use has a large impact on the regulatory pathway and thus the accumulated effort of bringing your device to the market. It is important that you consciously and carefully formalize the intended use and that you also develop the device according to the intended use.
The "Intended Use" plus "target market" will render into a product classification for each target market. FDA and EU regulations use different classification schemes. Generally speaking, the classification describes which and how much data you need to provide in order to get regulatory clearance. This can vary a lot between classifications.
Generally, the following concepts apply; the higher the risk the product poses to the user (patient, nurse, or other), the higher the classification. The higher the classification, the more work (including documentation) is required to prove that the product is safe.
If your device is in an area where you need to prove its effectiveness and reliability on living beings, you need to go through proper Clinical trials (see http://en.wikipedia.org/wiki/Clinical_trial ). Clinical trials are notoriously expensive. Therefore, it is crucial that you have a formal process in place, otherwise, the data will not be valid and you have to start all over.
Since patient safety is such a central concept in medical device development, the sooner you get familiar with Risk Management the better. There is simply no way around it. In order to launch your product, you have to have documented proof that your device is safe for patients and users. The normative standard to look for is ISO 14971 which describes how risks are identified and controlled. There are plenty of seminars and webinars available on risk management introduction and it is well spent time to visit one or more early on.
Medical device development documentation constitutes a large part of the total development effort. By learning how to apply Good Documentation Practices during the early phases to all your documents, you will feel more at ease with the necessary work methods when the “formal development” starts.
Switching from “non-formal” to “formal” development is not done in an instance. Setting up the development framework will take some time and you should plan accordingly. I have tried to introduce some of the most important medical device concepts in this text.
If you plan to stay in the Medical Device development field for some foreseeable time, regardless of role or occupation, these are concepts that you have to get familiar with. They will permeate your work. Even though they might not seem relevant to start-up teams, it is necessary to know these things in order to make the notoriously difficult transition from prototype to market-ready as smooth as possible.
If you have questions or want to know more about these things, please
Learn more about how Aligned Elements can help with achieving regulatory compliance
Request a live demo and let us show you how Aligned Elements can manage your documentation
{fastsocialshare}
I say "IEC 62304 Software Safety Classification", you say "A, B, C".
If you have worked with IEC 62304, you have (probably) also faced the challenge of assigning Software Safety Classifications to the Software Items. It has to do with associating a risk level to your software items in order to assess how careful you need to be when you document, develop and test them.
Now, if you work with medical device development, it has probably dawned on you that you are already doing risk management. Lots of risk management. All sorts of risk management. Is it possible to use the existing risk assessments when doing our IEC 62304 work? Yes, I think so.
When a software designer/architect constructs the software architecture needed for IEC 62304, there are lots of approaches to choose from. The norm is explicitly ambiguous on this point. In short, it is up to you to pick an approach. It is however not uncommon for the software designer to use established modeling techniques such as UML, Object-oriented design, multi-layered design, SAAS, etc. which in most cases results in a number of boxes (the software items) with names like:
So are we supposed to perform a risk assessment of these boxes now?
What strikes you immediately is the insight that these boxes in themselves do not "have" risk. Why? Because we have not defined the context in which they are used. Software risk emanates from how we use the software i.e. in which functional aspect we use the software items.
The guys of Certified Compliance Solution have solved this in a really neat way using what they call "The Matrix Model for Software Safety Classification". It is well summarized in the picture below.

The CCE people have correctly identified that functional flows cut across the software architecture.
In their model, Software Items are listed on the horizontal axis, and User stories (or Epics, Use Cases, Work Flows, Specification, or whatever you want to call them) are listed on the vertical axis. Now, risk assessments for User Stories are something we do every day so that part should be pretty straight-forward. Once that is completed, we can deduct a classification for each User Story based on this risk assessment (more about that later).
The Software Designer can now point out the Software Items that implement our User Story in the software. After we have done this for all User Stories, the software Safety Classification for a Software Item is simply the highest risk of its aggregated use, which is summarized in the bottom line "Overall SW Item Safety Class".
Isn't it beautiful?
Let me show you how this is implemented in Aligned Elements.
First, I introduce the Document Object type "Use Case" which will represent the user stories. Then I set up Document Object types for Software System, Software Item, Software Unit, and SOUP which all have a Software Safety Class attribute being A, B, or C, with the default value C.
I can then proceed to document the Use Cases and structure the Software Architecture separately.
Now, time for risk assessment. I will use the integrated Preliminary Hazard Analysis (PHA) to assess my Use Cases. When working out the risks using the PHA method, I am requested to define the possible Cause(s) for each risk. The Cause has a severity attribute and I will use this severity to calculate the Software Safety classification. If the Cause is emanating from the software, I further tag the Cause originator as "Software".
I go back to the Software Item definition and map the values A, B, and C to a Severity range in the Cause i.e. a Severity from 1-3 maps to Class A, Severity 3-5 maps to Class B, etc.
To tie it all together, I trace the Use Cases to the PHA risks and (here comes the trick) Software Items to the software originated Causes.
I run an Inconsistency analysis on the Software Items and get a report on how well the Software Item Safety classifications match the risk assessment, including suggestions on what the Software Safety Classification ought to be based on the associated risks.
Simple, transparent, and consistent.
So we have a range of benefits from this approach:
Once again we have shown the benefit of integrating all Design Control Items in a single system.
The sum is truly greater than its parts.
{fastsocialshare}