- Icon: <i class="fa-solid fa-file-contract"></i>
-
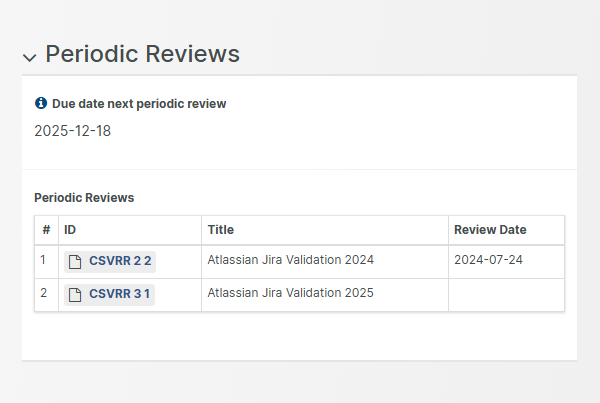
A complete set of SOPs to kick-start your road to market approval
The Aligned eQMS is delivered with a complete set of 32 Standard Operating Procedures and 107 templates specifically developed for ISO 13485 certification.
This set, covering all types of medical devices, is designed to make you fully compliant with MDR, IVDR, ISO 13485, FDA 21 CFR 820, ISO 14971 and IEC 62304.
Use the Aligned eQMS Word Add-In to make organization specific changes or complement this set by uploading your own MS Office documents.
-
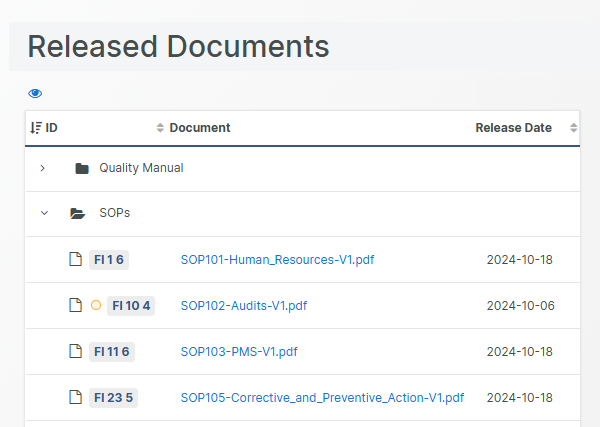
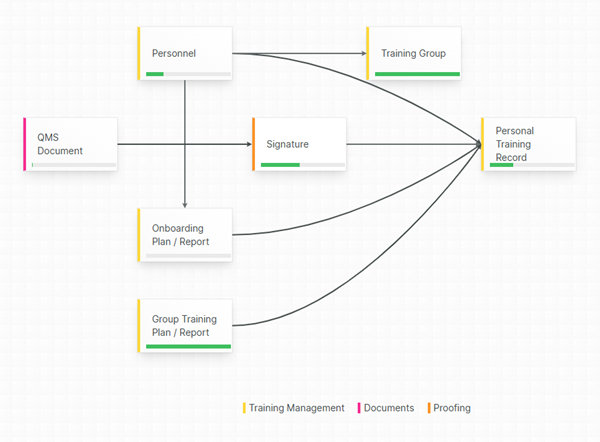
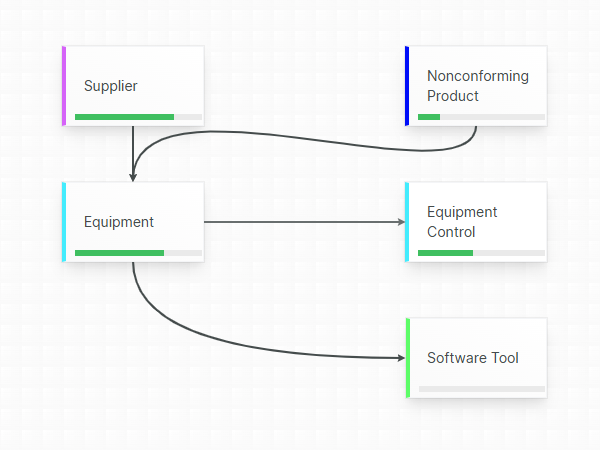
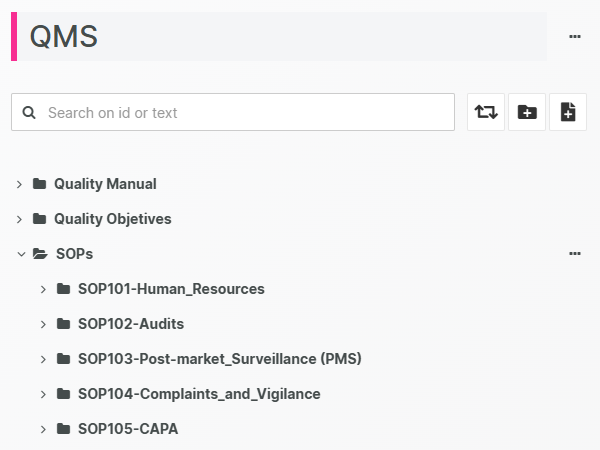
Collaborative, centralized Document Management
A single source-of-truth for your quality team with zero document duplicates, always up-to-date.
Eliminate documentation confusion by relying on a centralized hub for your QMS Documents, protected by secure and permission-based access.
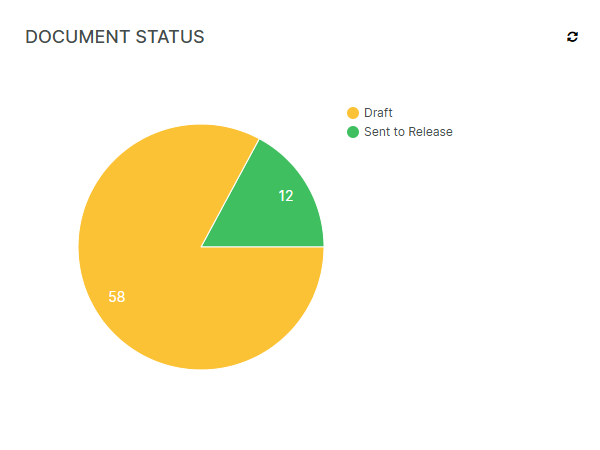
A clear separation between formally released documents and documents currently being drafted (or being changed, reviewed or in the middle of an approval cycle) minimizes the risk of mistaking a non-released document for a released one.
-
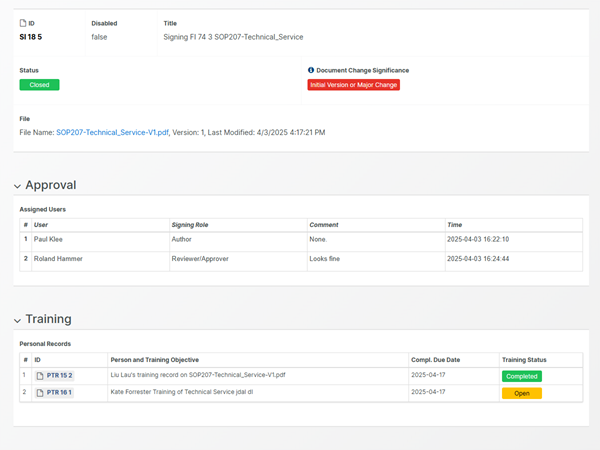
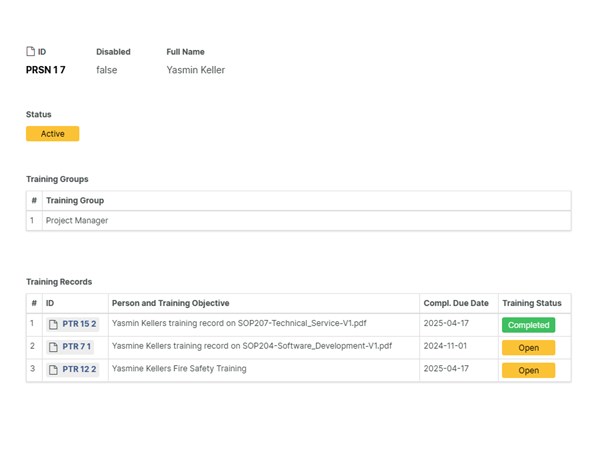
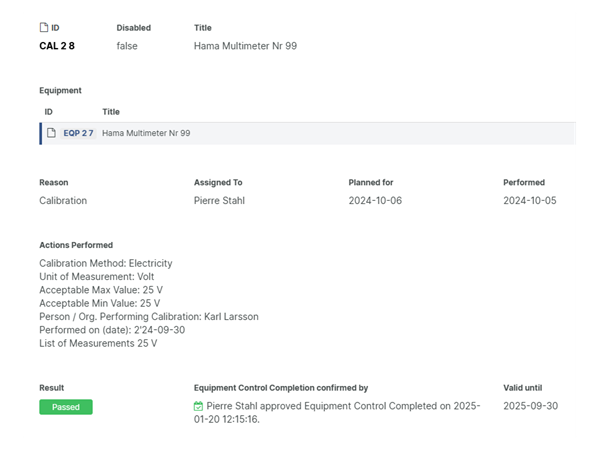
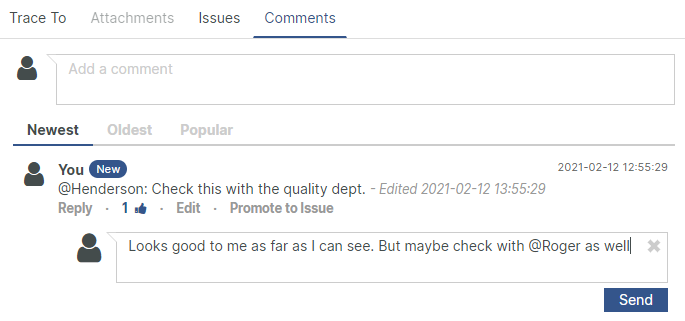
Digitalized review, approval and signing
With Aligned's FDA 21 CFR Part 11 compliant signatures, routing and approval of documents is automated.
Effective sign-offs, using sequential or parallel approval signature workflows. Optimize approval speeds by assigning signatures to roles instead of individual persons, using a "first-come, first-serve" approach.
Stay in the loop with automatic email alerts and designated "Assign to me"-dashboards, updated as soon as a signing action has been routed to you.
-
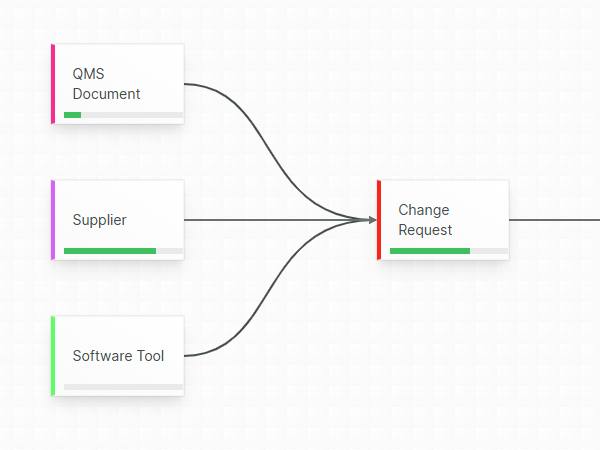
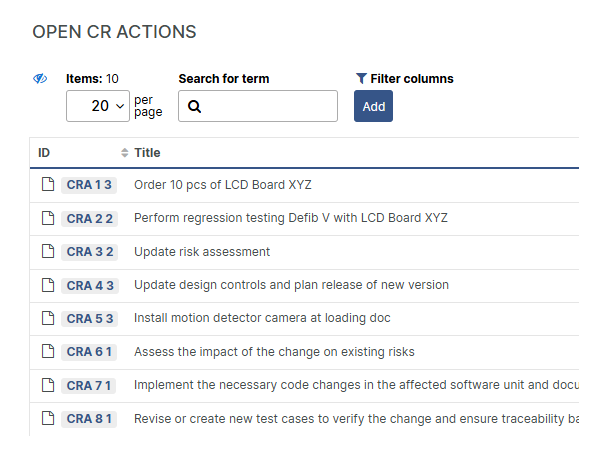
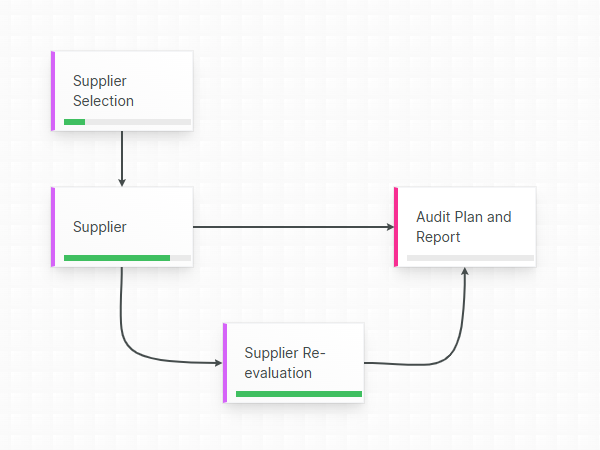
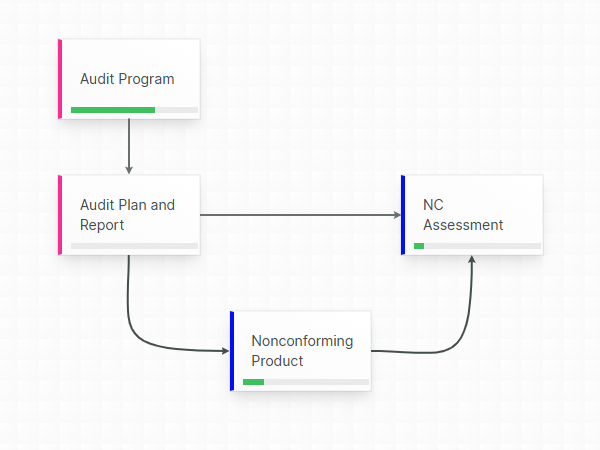
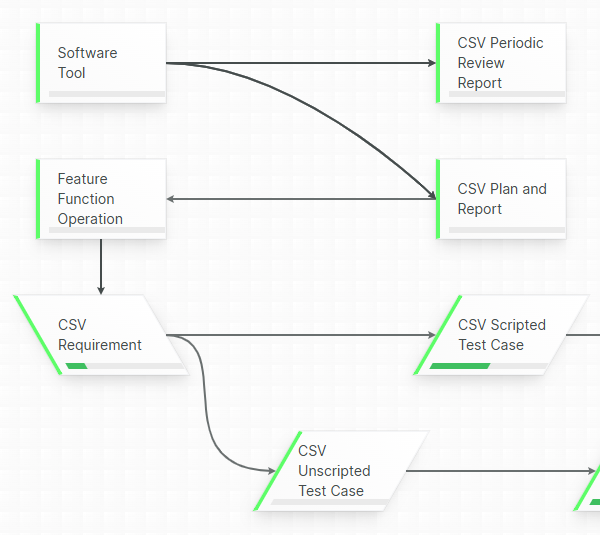
Confident, compliant, audit-ready
Accountability guaranteed by mandatory revision control, where each document change is stored into a chronological an unambiguous audit trail.
Aligned eQMS gives you the necessary mechanisms to guarantee that only released templates are used in your Technical Files.
Respond faster to audit and inspection demands by swiftly and accurately locating the relevant released document, leveraging the built-in full text-search, powerful filtering and extensive query possibilities.
Ready to get started?
Try the Aligned eQMS 30 days for free!
Genuine Voices, Tangible Results
Genuine Voices, Tangible Results