{fastsocialshare}
In this recent New in Aligned Elements V2.5 SP 4 Webinar we presented news and improvements including:
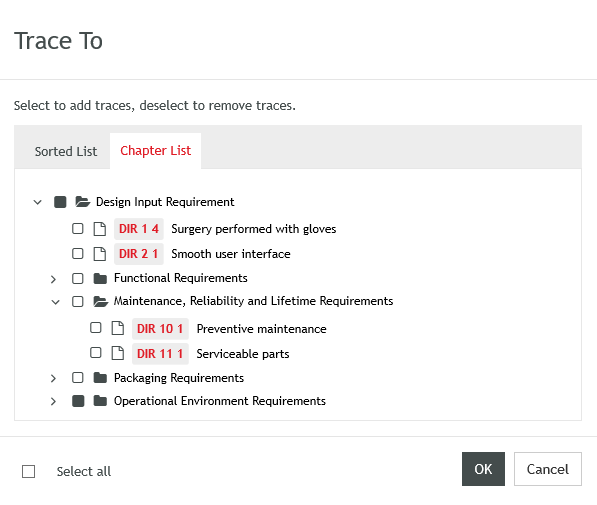
- A new UX experience
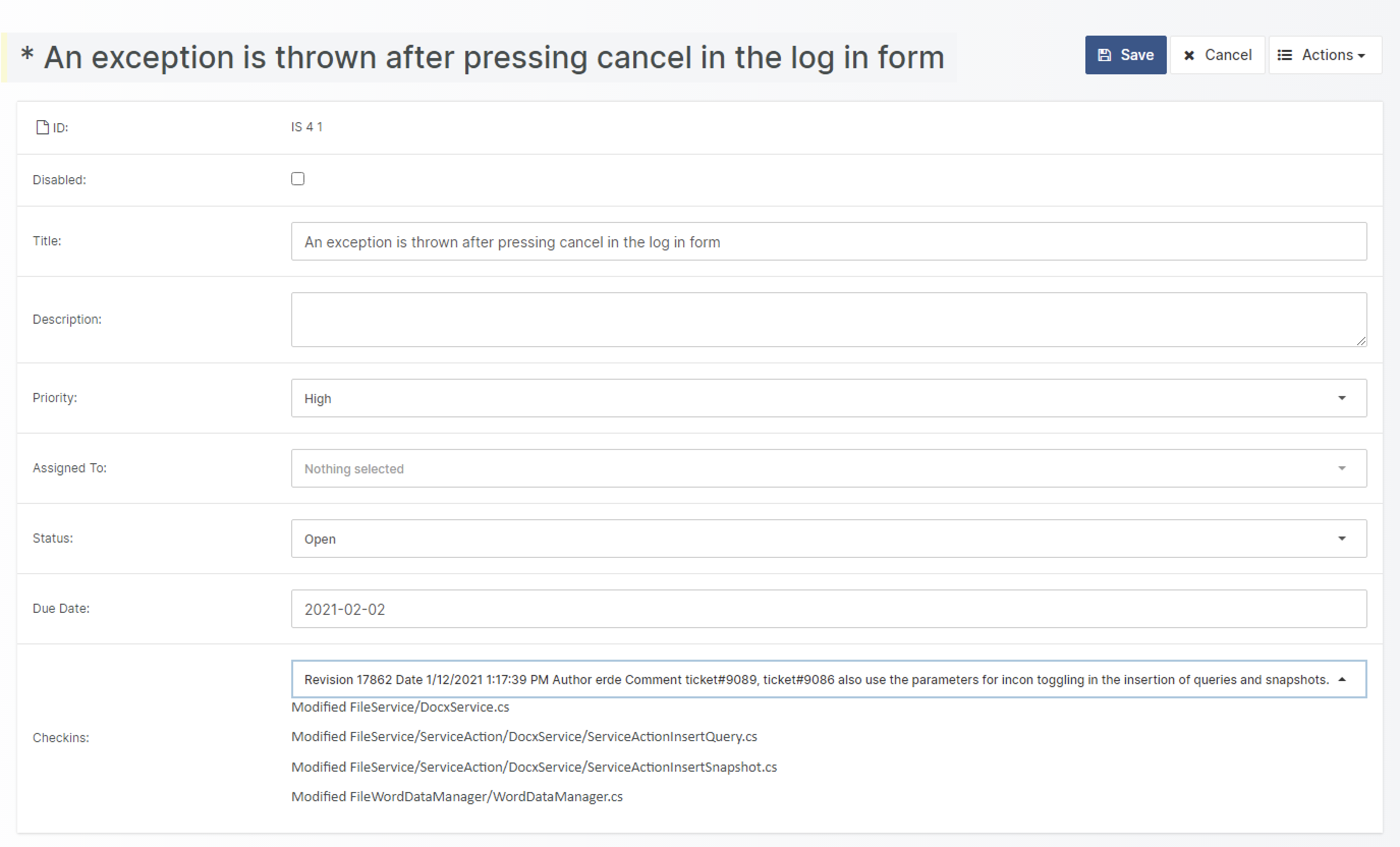
- Enhanced Review and Feedback Options
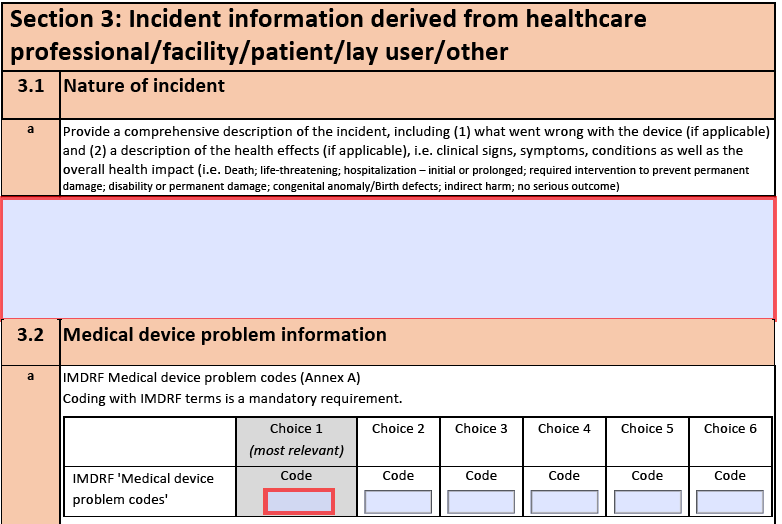
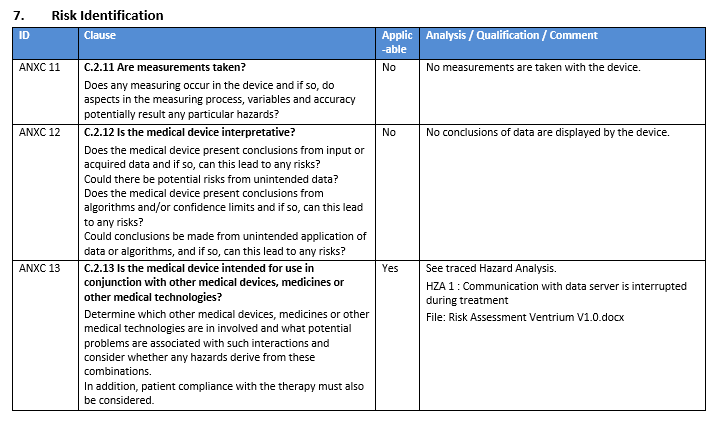
- Highlighting Inconsistent Items in Word Documents
- Integration with GitLab and SVN
- Centralized User Management
...and much more.
The recording is available below.
Enjoy!