The Aligned Elements IEC 82304 configuration is a superset of the IEC 62304 configuration. It expands the IEC 62304 configuration with Product Use Requirements and Product Validation Tests. It has been tuned to automatically take care of most of the involved quality checks, making sure that the required tasks and actions are sufficiently covered.
The Aligned Elements IEC 82304 configuration contains:
- Pre-configured templates using IEC 82304 standard naming conventions
- Software Safety Classification automatically based on risk analysis results
- Numerous quality checks for consistency verification
- Pre-configured Reviews and checkpoints according to IEC 82304 and IEC 62304 stipulations
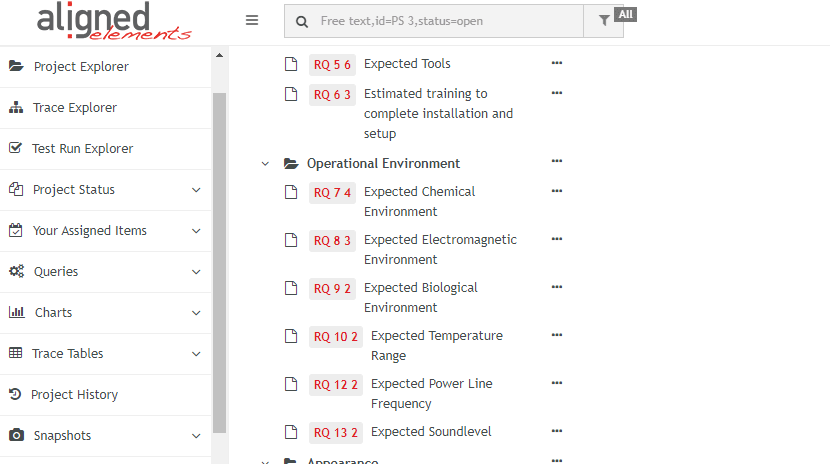
- Pre-configured Trace Tables based on the IEC 82304 and IEC 62304 requirements
- A set of document templates being a great starting point for your documentation
- 48 importable Product Use Requirements for Accompanying Documentation from IEC 82304
The Aligned Elements IEC 82304 supports documentation management of:
- Product Use Requirements
- Product Validation Tests and Results
- System and Software Requirements
- Software Architecture building blocks(Software Items, Units, SOUPs, and segregations)
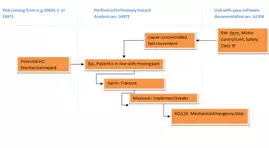
- Risk Management using a Preliminary Hazard Analysis technique (listed in ISO 14971)
- Software Verification (Unit, Integration and System testing)
- Change and configuration management (Problem Reports and Change Management)
Note! The Aligned Elements IEC 82304 configuration is a significantly large extension to apply.
To get the proper understanding of the extension aspects, we strongly recommend that you book a live demonstration of the Aligned Elements IEC 82304 configuration, please