With the Aligned Elements V2.4 Service Pack 1 (2.4.132.12658) we have fixed a small number of bugs.
This service pack mainly addresses known problems with Word 2010 and Word 2007 from Aligned Elements V2.4.
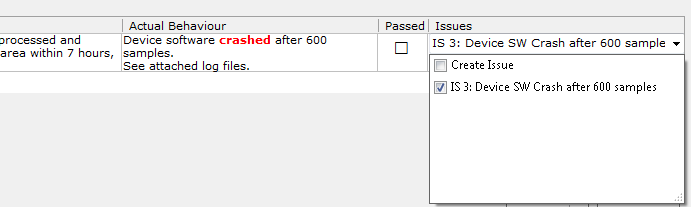
It also provides fixes to the integration mechanisms with Countersoft Gemini and the usability of Test Runs.
If your team is using Word 2010 or Word 2007, you are recommended to upgrade to this service pack.